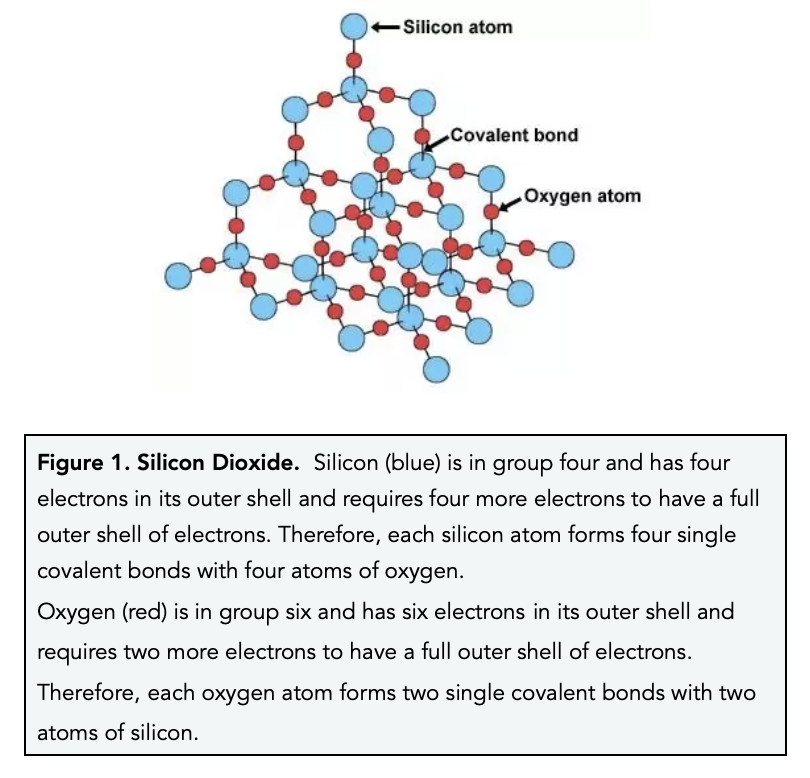
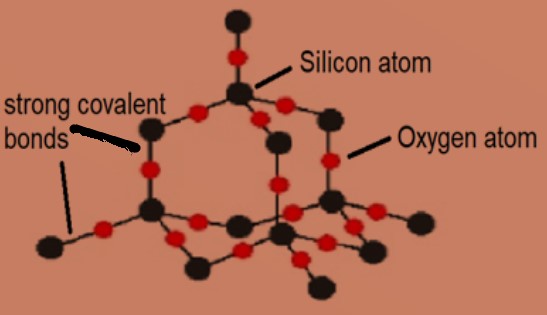
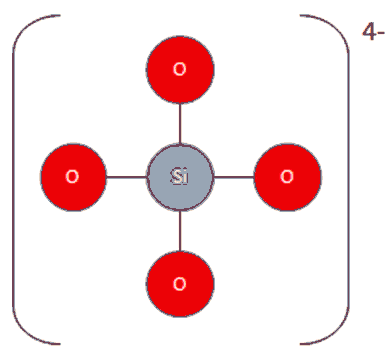
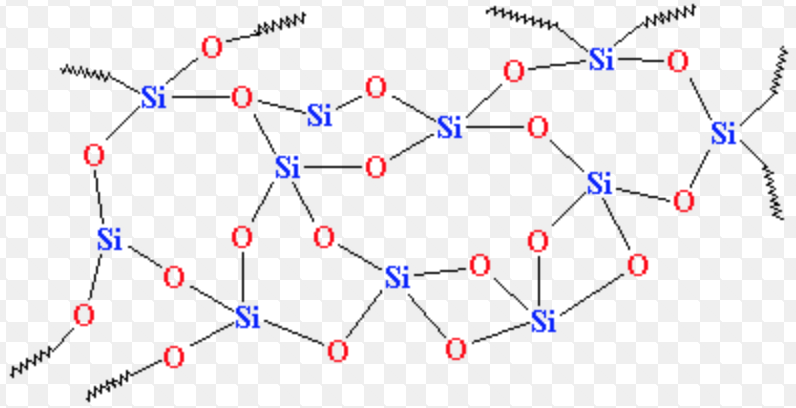
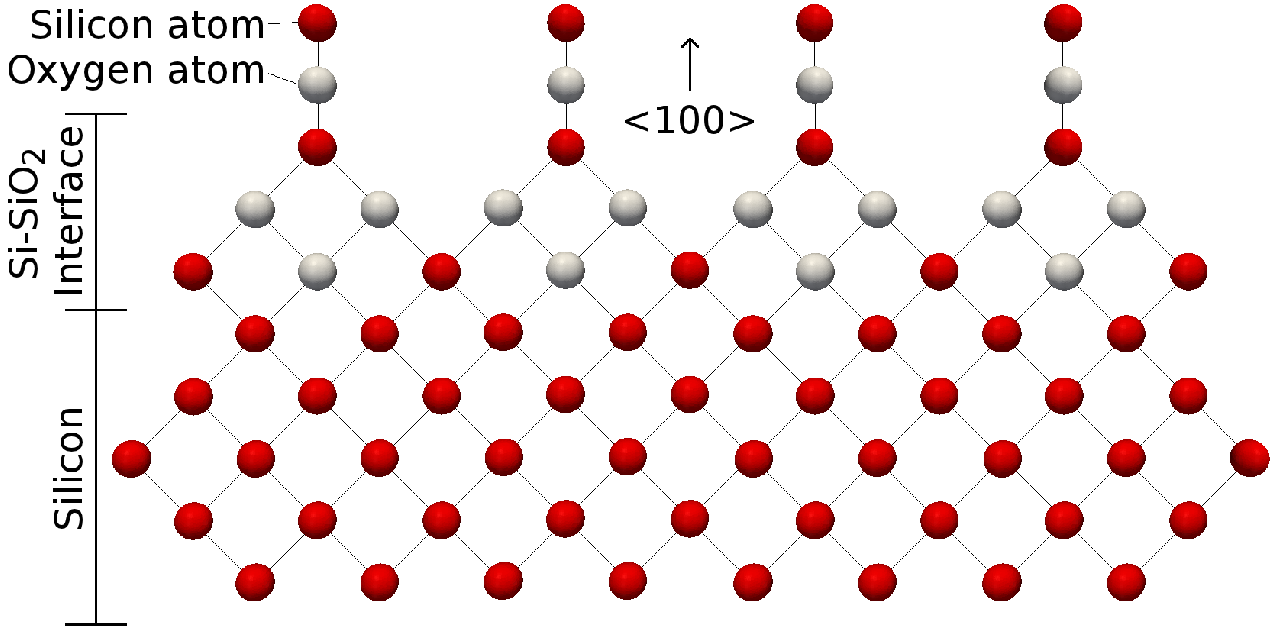
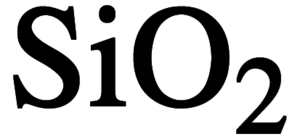Why does silicon(IV) oxide have the formula Si02 but a structure in which each silicon atom is bonded to four oxygen atoms? - Quora
Silicate Chemistry Silicon Dioxide Lewis Structure Tetrahedron, PNG, 960x511px, Silicate, Area, Black, Black And White, Brand
![Figure 1 from Structure, stability, and dissociation of small ionic silicon oxide clusters [SiO(n)+ (n = 3, 4)]: insight from density functional and topological exploration. | Semantic Scholar Figure 1 from Structure, stability, and dissociation of small ionic silicon oxide clusters [SiO(n)+ (n = 3, 4)]: insight from density functional and topological exploration. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7cfd658ee511b9c786e1124b5d06ec4905dc4443/5-Figure1-1.png)