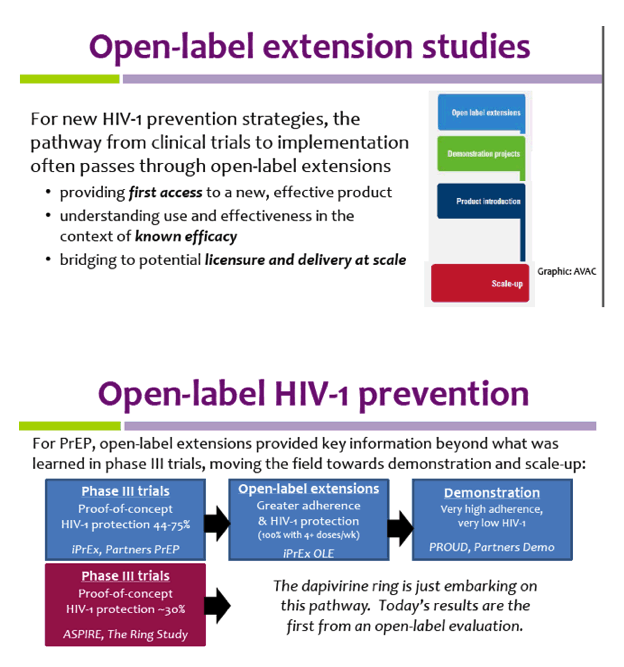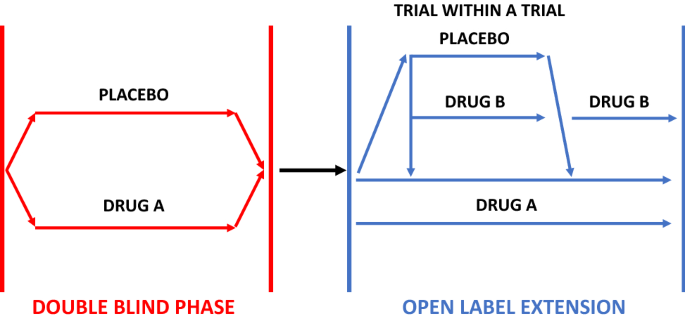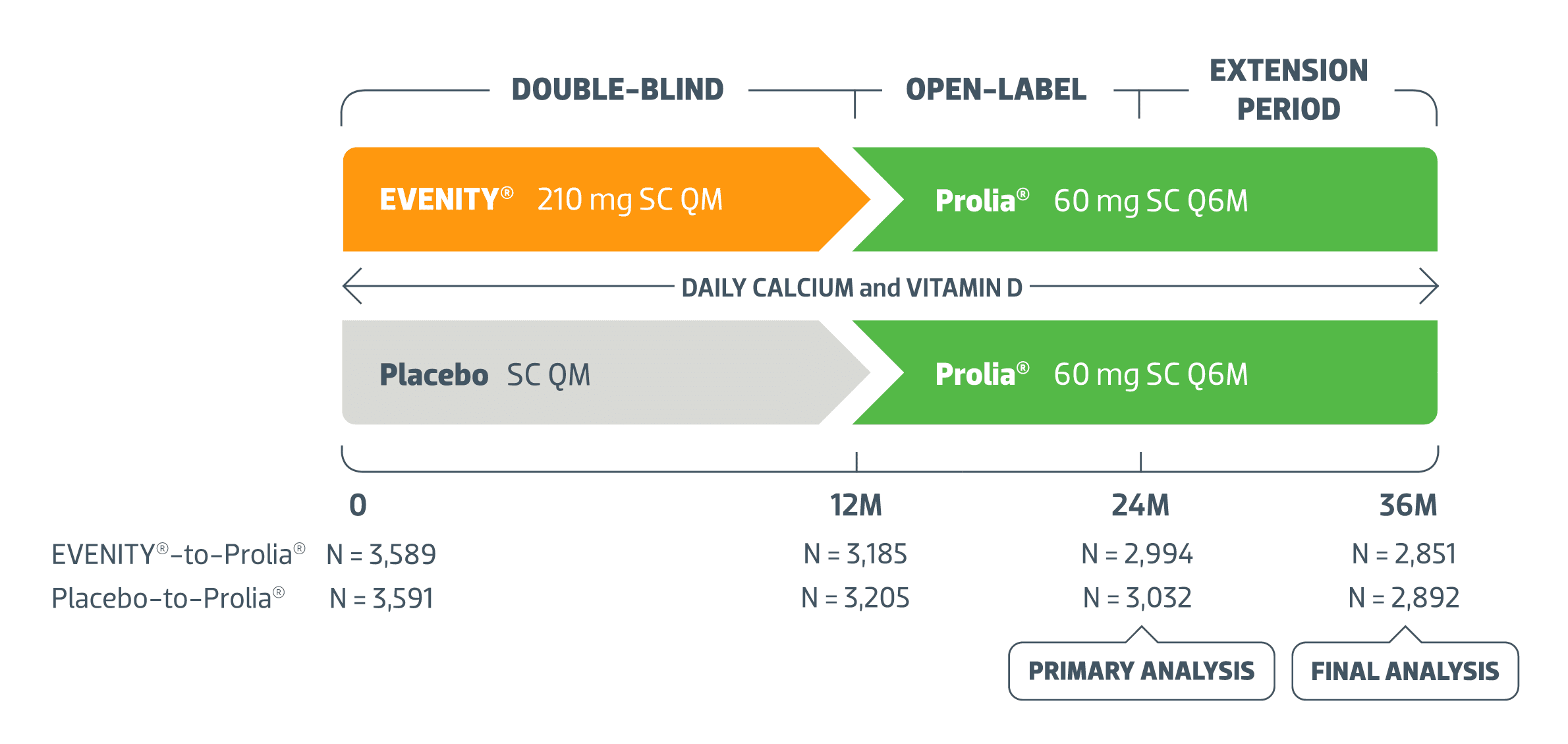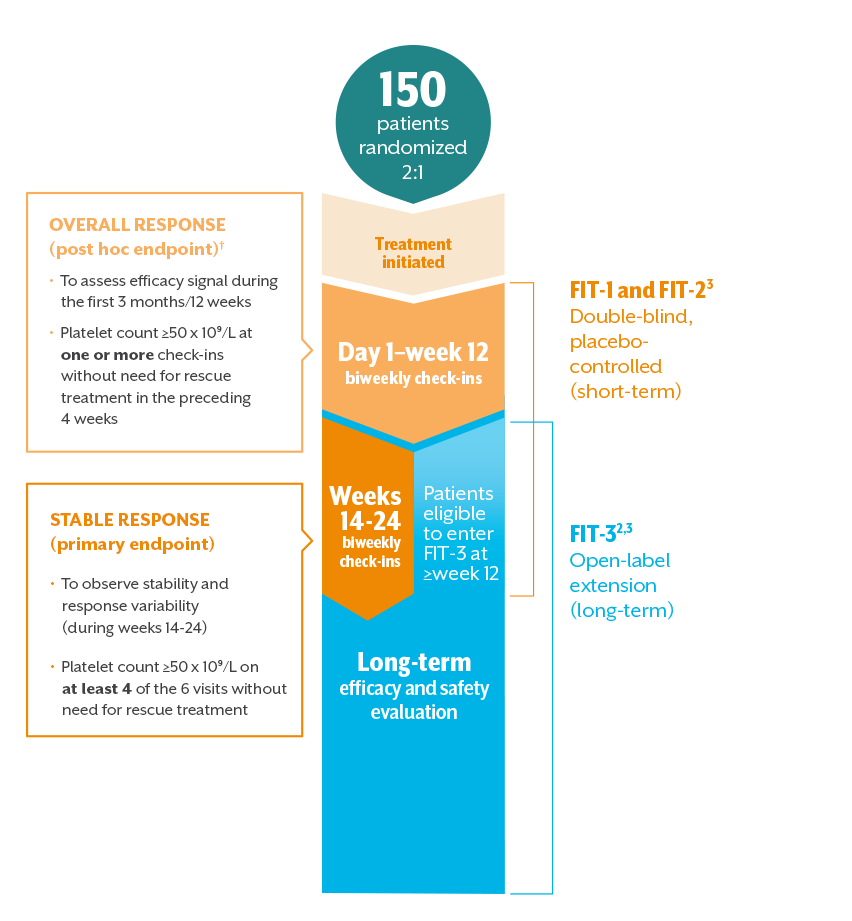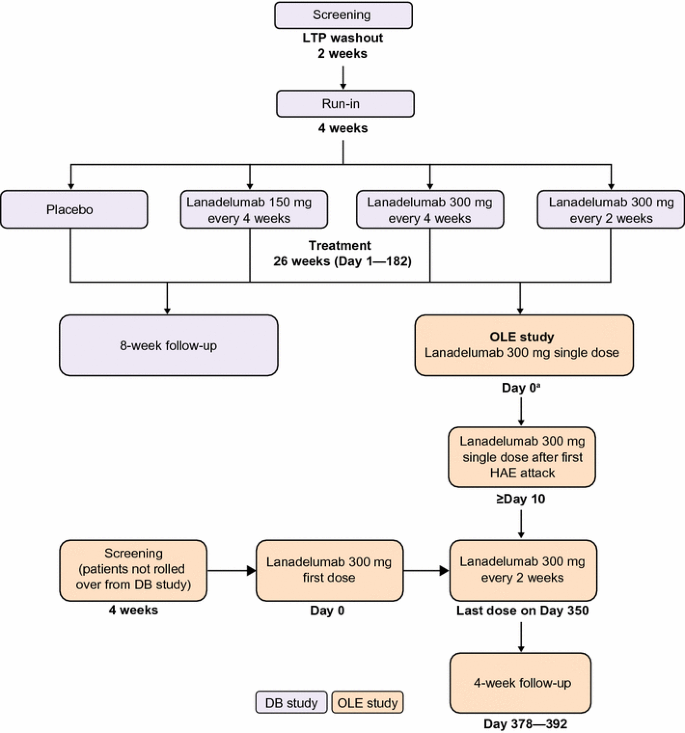
An open-label study to evaluate the long-term safety and efficacy of lanadelumab for prevention of attacks in hereditary angioedema: design of the HELP study extension | Clinical and Translational Allergy | Full

Study design schematic for the open-label extension study. AE, adverse... | Download Scientific Diagram
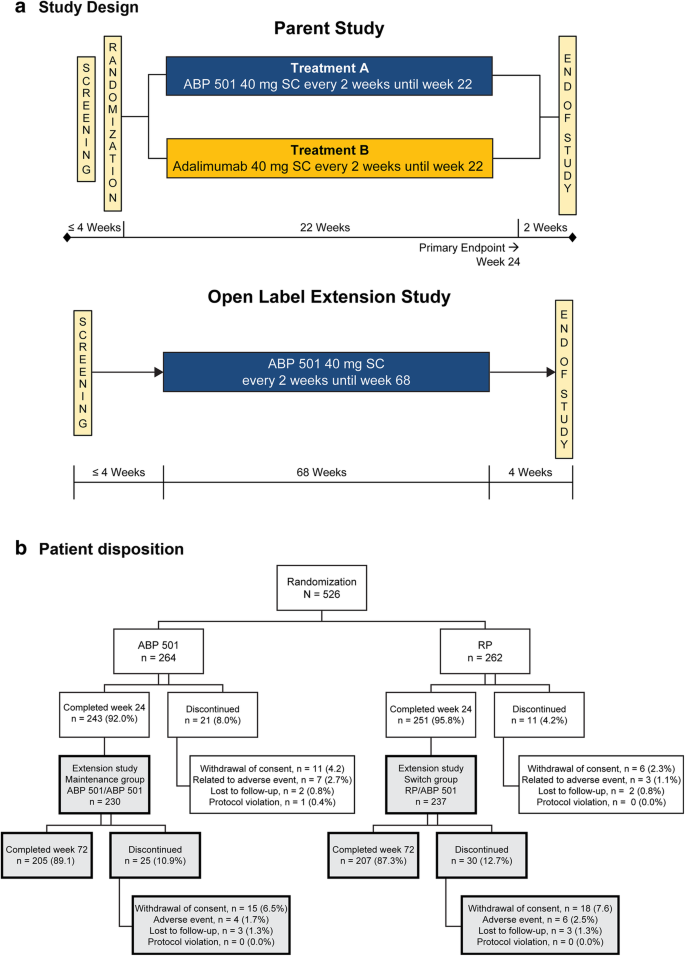
An open-label extension study to demonstrate long-term safety and efficacy of ABP 501 in patients with rheumatoid arthritis | Arthritis Research & Therapy | Full Text

Uncontrolled Extensions of Clinical Trials and the Use of External Controls—Scoping Opportunities and Methods - Wang - 2022 - Clinical Pharmacology & Therapeutics - Wiley Online Library

Summary of Other Studies, Open-Label Extensions - Clinical Review Report: Telotristat (Xermelo) - NCBI Bookshelf
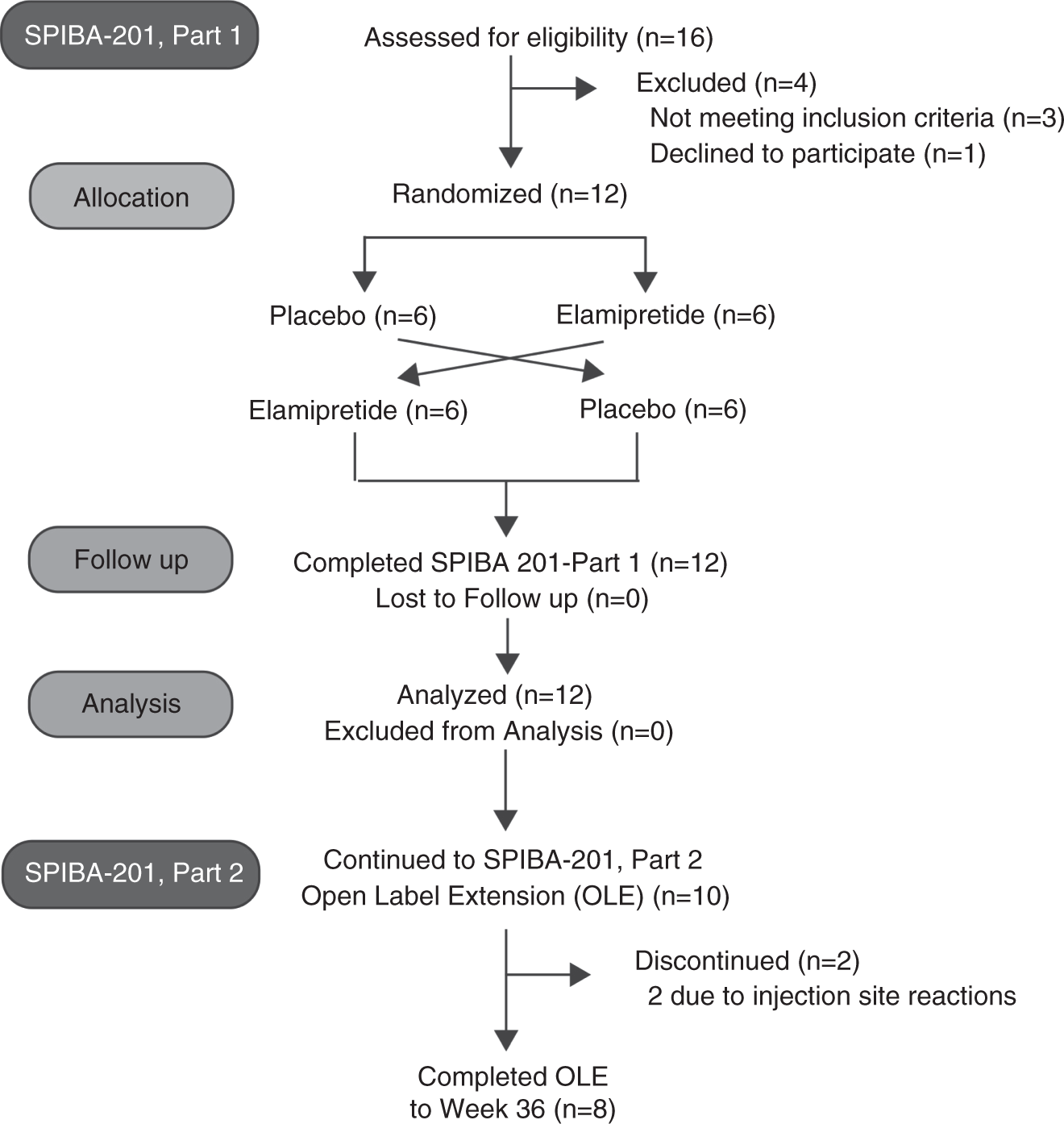
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine

Safety and efficacy of rituximab in systemic sclerosis (DESIRES): open-label extension of a double-blind, investigators-initiated, randomised, placebo-controlled trial - The Lancet Rheumatology

A 6-month open-label extension study of the safety and efficacy of subcutaneous belimumab in patients with systemic lupus erythematosus - A Doria, D Bass, A Schwarting, A Hammer, D Gordon, M Scheinberg,

Flow chart showing the study design of the open-label extension phase... | Download Scientific Diagram