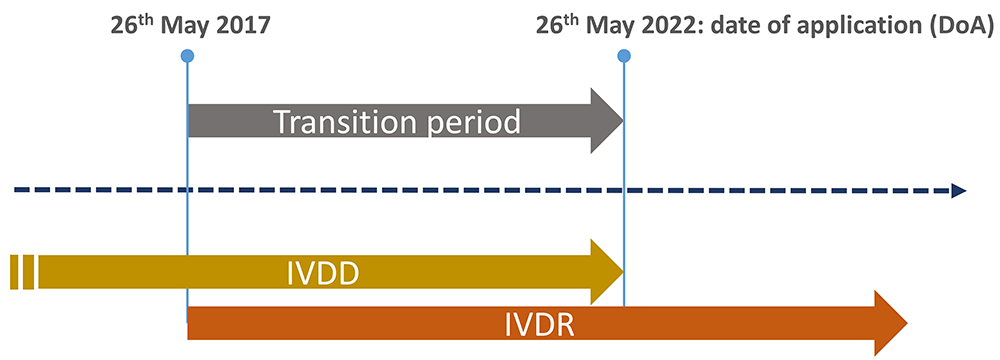
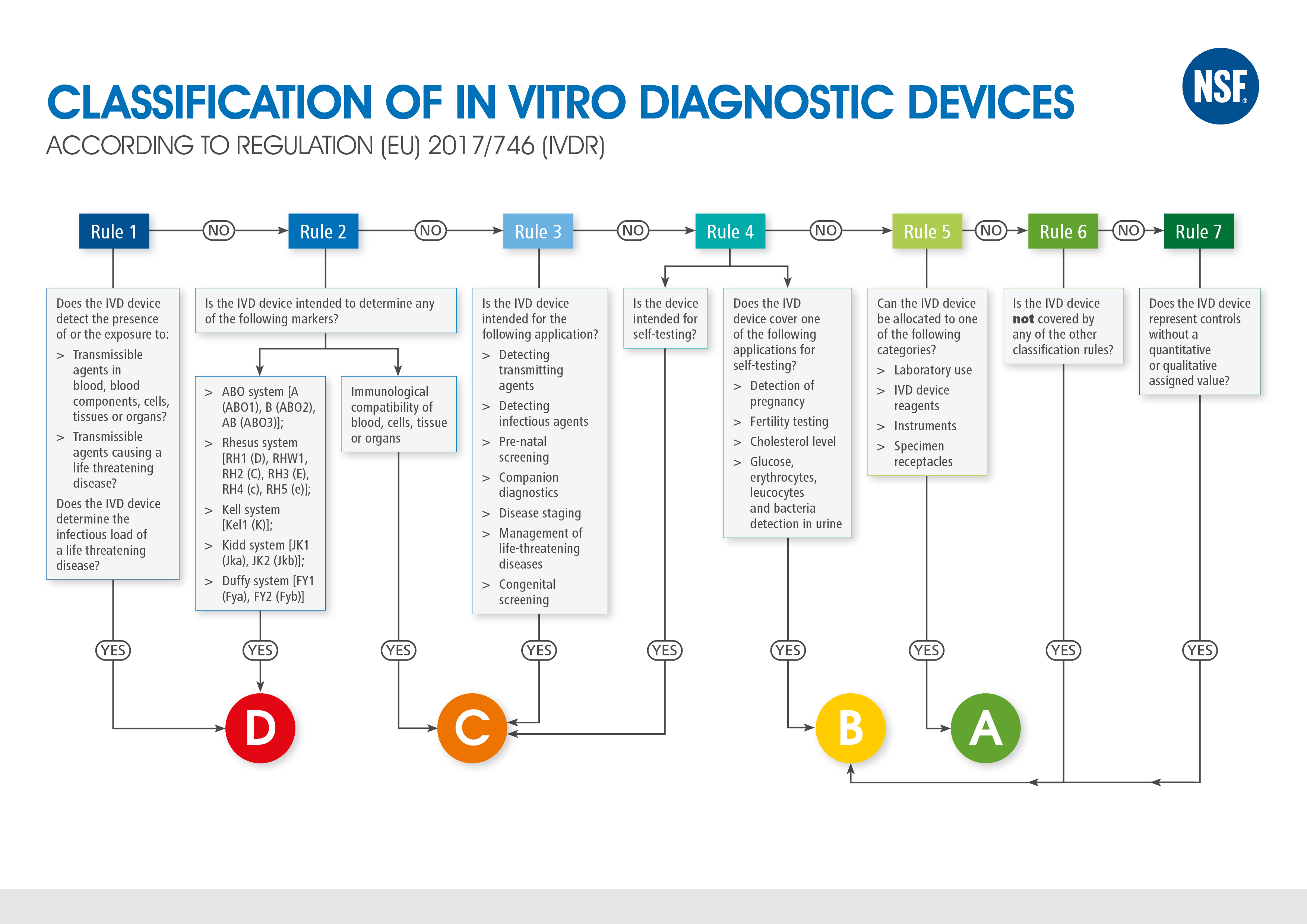
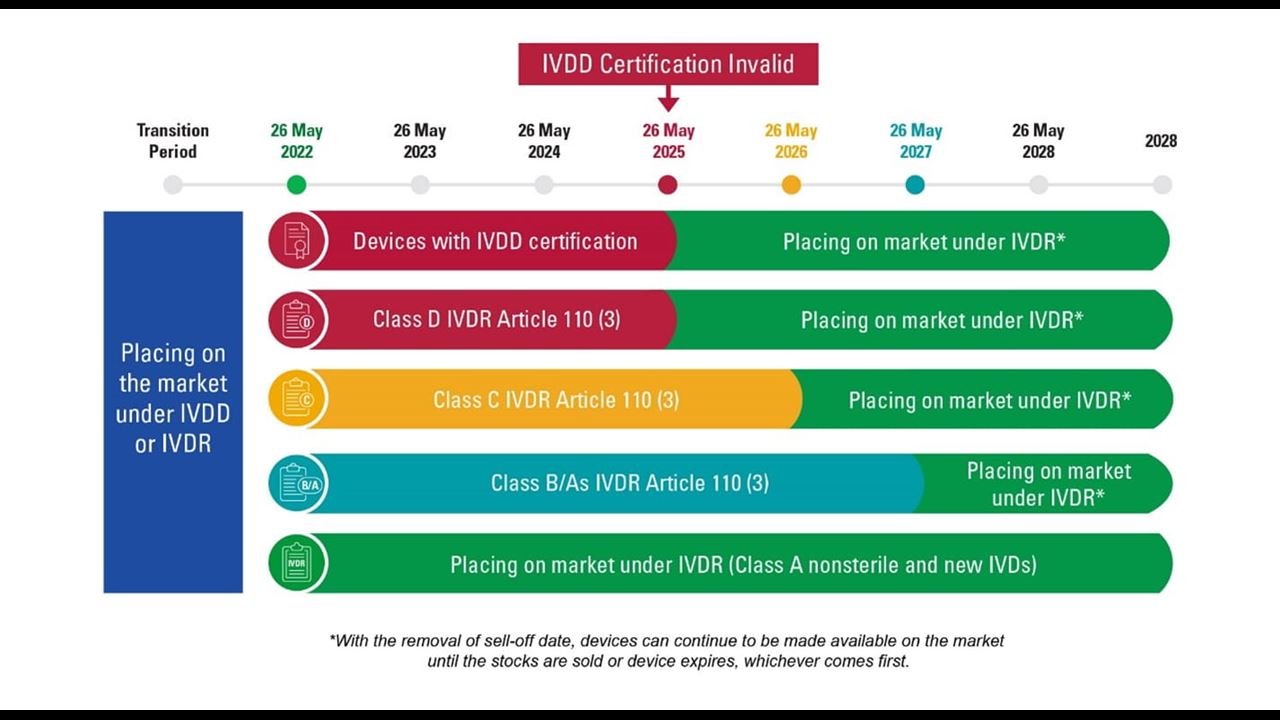
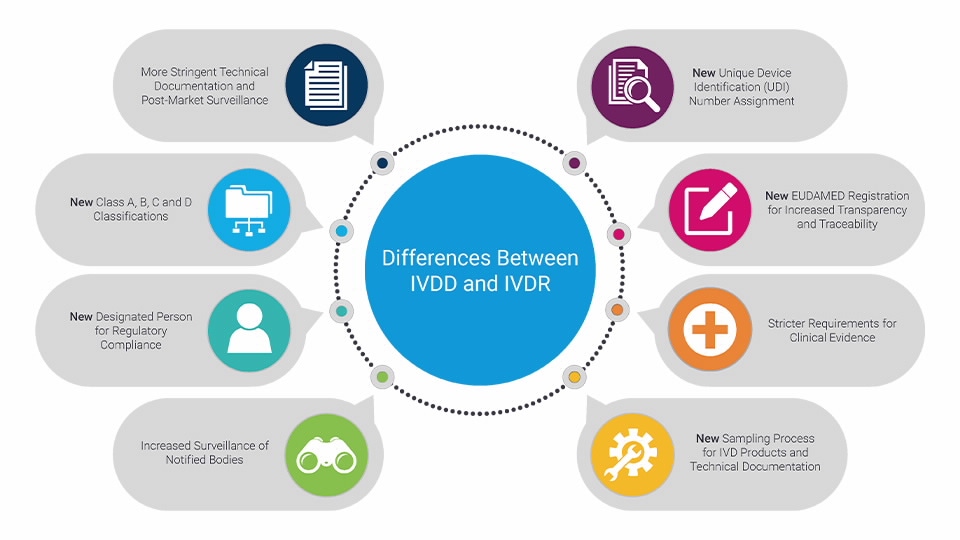
TÜV SÜD @TUVSUD emite el primer certificado con el reglamento IVDR a un producto clase B – Enhorabuena!!

5 key implications from the new EU In-vitro Diagnostics Regulation — (Digital) Diagnostics Market Access & Policy Consulting based in Switzerland

In vitro diagnostic software: Novelties introduced by Regulation (EU) 2017/746 - GMED Medical Device Certification

Medical device regulation in Europe – what is changing and how can I become more involved? - EuroIntervention