How to Balance Fe + H2SO4 = FeSO4 + Fe2(SO4)3 + H2O + SO2 (Iron + Concentrated Sulfuric acid) - YouTube
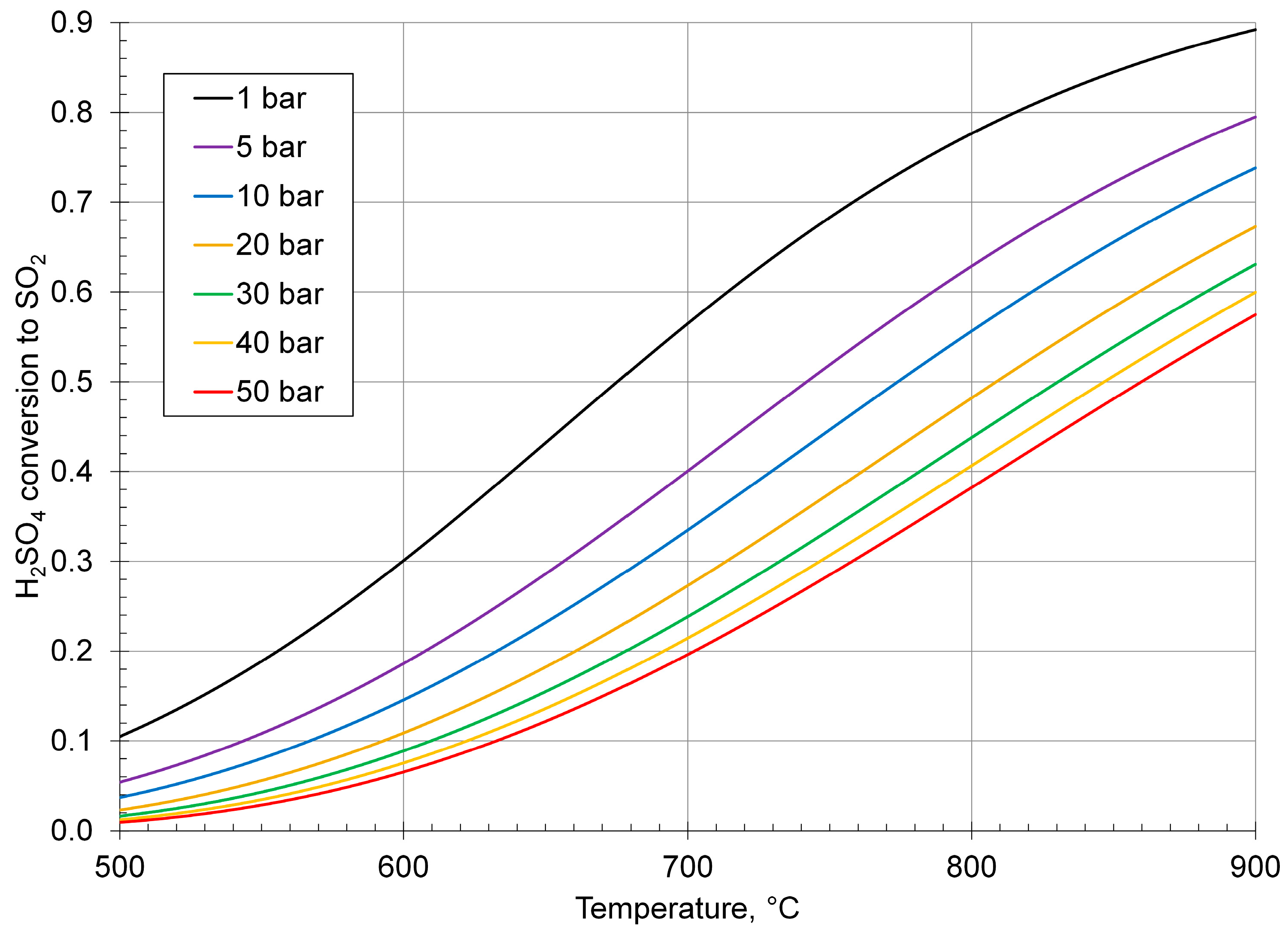
Processes | Free Full-Text | Review of Sulfuric Acid Decomposition Processes for Sulfur-Based Thermochemical Hydrogen Production Cycles

How to Balance Fe + H2SO4 = FeSO4 + Fe2(SO4)3 + H2O + SO2 (Iron + Concentrated Sulfuric acid) - YouTube

How many kilograms of pure H2SO4 could be obtained from 1 kg of iron pyrites (FeS2) according to the following reactions ? 4FeS2 + 11O2 → 2Fe2O3 + 8SO2 2SO2 + O2 → 2SO3 SO3 + H2O → H2SO4
![When conc. H2SO4 was treated with K4 [ Fe (CN)6 ] , CO gas was evolved. By mistake, somebody used dilute H2SO4 instead of conc. H2SO4 then the gas evolved was: When conc. H2SO4 was treated with K4 [ Fe (CN)6 ] , CO gas was evolved. By mistake, somebody used dilute H2SO4 instead of conc. H2SO4 then the gas evolved was:](https://i.ytimg.com/vi/dU3X9OSOjt8/maxresdefault.jpg)
When conc. H2SO4 was treated with K4 [ Fe (CN)6 ] , CO gas was evolved. By mistake, somebody used dilute H2SO4 instead of conc. H2SO4 then the gas evolved was:

Effect of H2SO4 concentration on iron and titanium leaching from the... | Download Scientific Diagram
IBP1218_09 CORROSION BY CONCENTRATED SULFURIC ACID IN CARBON STEEL PIPES AND TANKS – STATE OF THE ART Zehbour Panossian , Neus

Doubt: WHAT HAPPENS WHEN K4[Fe{CN}6} REACTS WITH CONC. AND DILUTE H2SO4? Chapter: The p-Block Elements (XII) - Subject: Chemistry - Course: NEET Course - Complete Syllabus
i) Fe + dil. H2SO4 (ii) Benzene + H2SO2(conc.) (iii) PCl5 + H2SO4 - Sarthaks eConnect | Largest Online Education Community