a) Concentration vs. time profile and (b) rate vs. time profile for... | Download Scientific Diagram

Calculating Average and Instantaneous Reaction Rate from a Graph of Concentration Versus Time Practice | Chemistry Practice Problems | Study.com

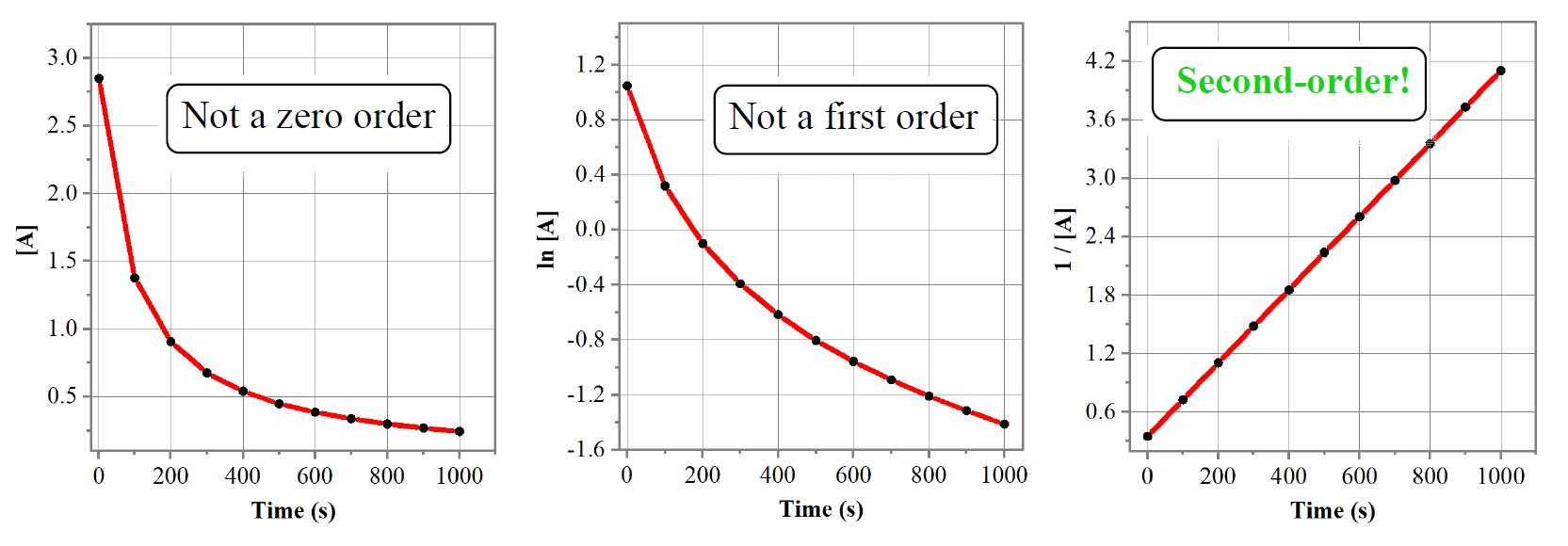
Analyse the given graph drawn between the concentration of reactant vs time Predict the order of the reaction
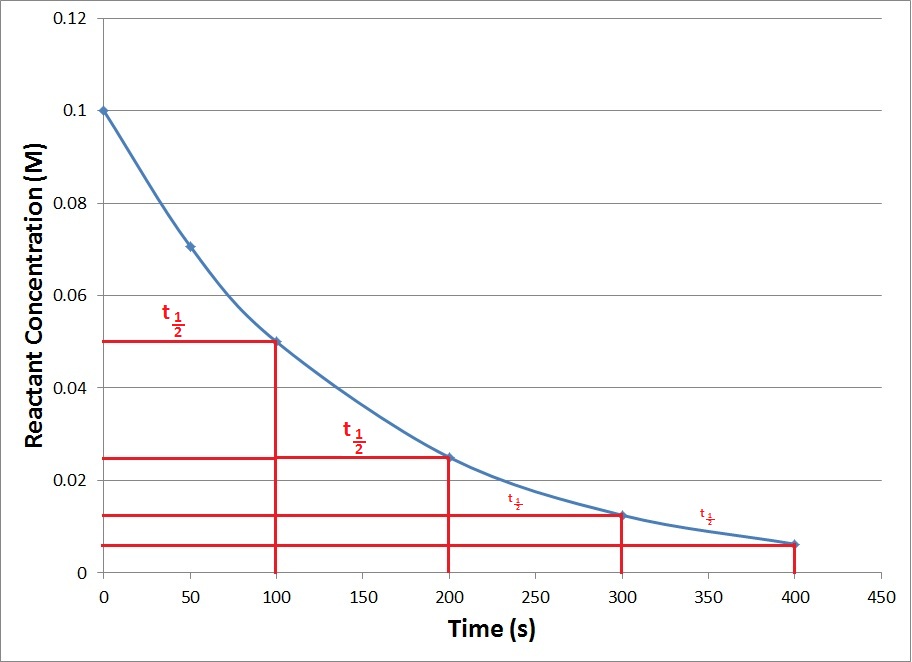
Concentration–Time Relationships: Integrated Rate Laws – Introductory Chemistry – 1st Canadian Edition

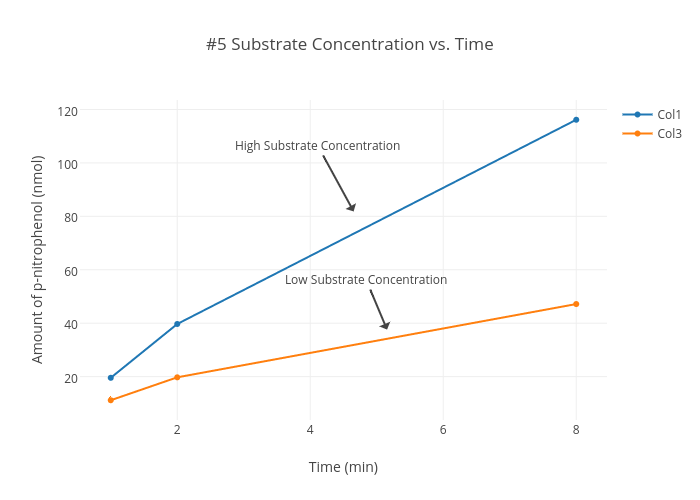
For a reaction A → Product, graph of concentration of reactant versus times is given. The rate (in Ms^-1 ) of reaction after 53.34 second from starting is:

Calculating Average and Instantaneous Reaction Rate from a Graph of Concentration Versus Time | Chemistry | Study.com

Comparison of log concentration versus time profile Acupan versus Inza... | Download Scientific Diagram

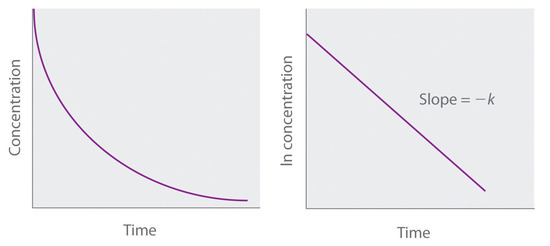
A: concentration versus time graph for a first-order reaction. b: graph... | Download Scientific Diagram
![Concentrations of A, B and C vs time are graphed for the reaction A ⟶ 3 B + C. The rate of the reaction is defined as r = k [ A ] Concentrations of A, B and C vs time are graphed for the reaction A ⟶ 3 B + C. The rate of the reaction is defined as r = k [ A ]](https://s3-us-west-2.amazonaws.com/infinitestudent-images/ckeditor_assets/pictures/425159/original_JEE_ADV05.png)
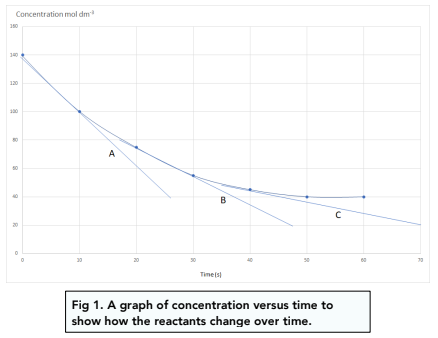
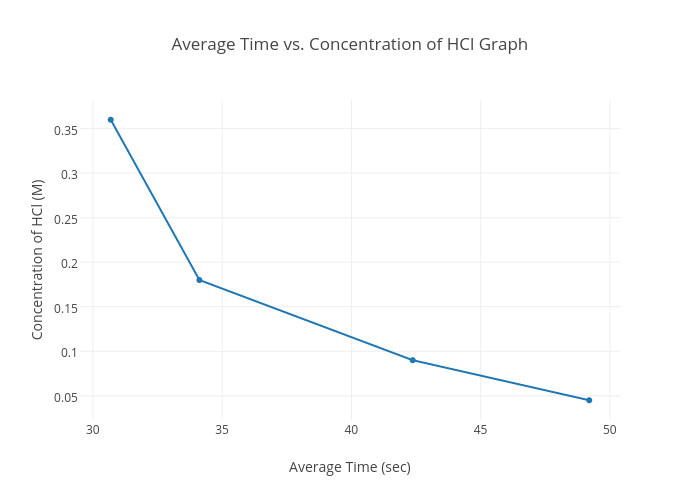
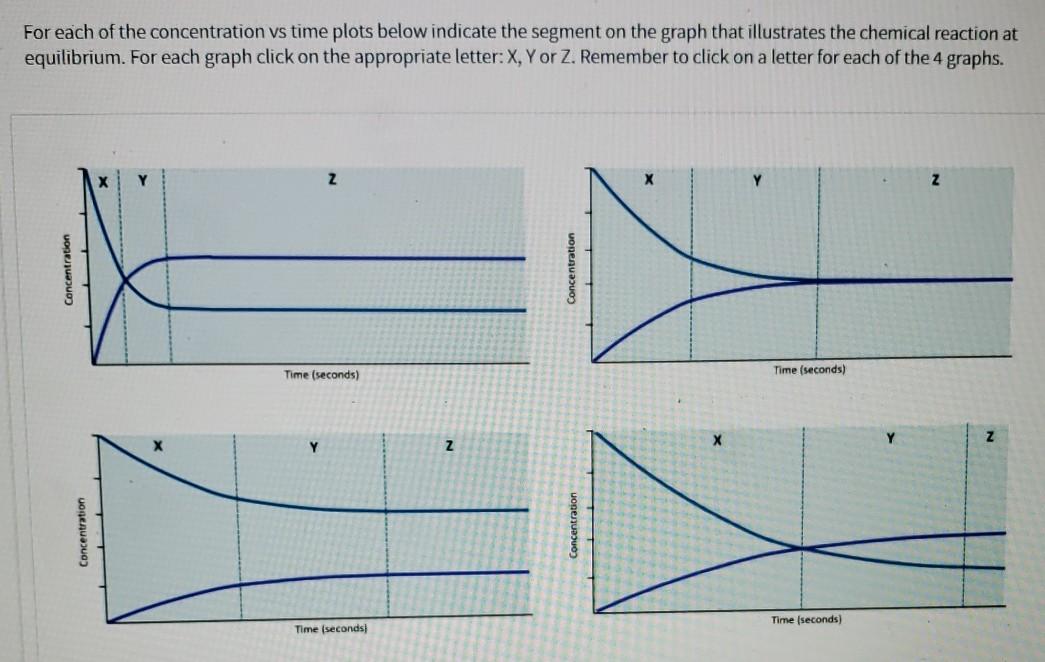
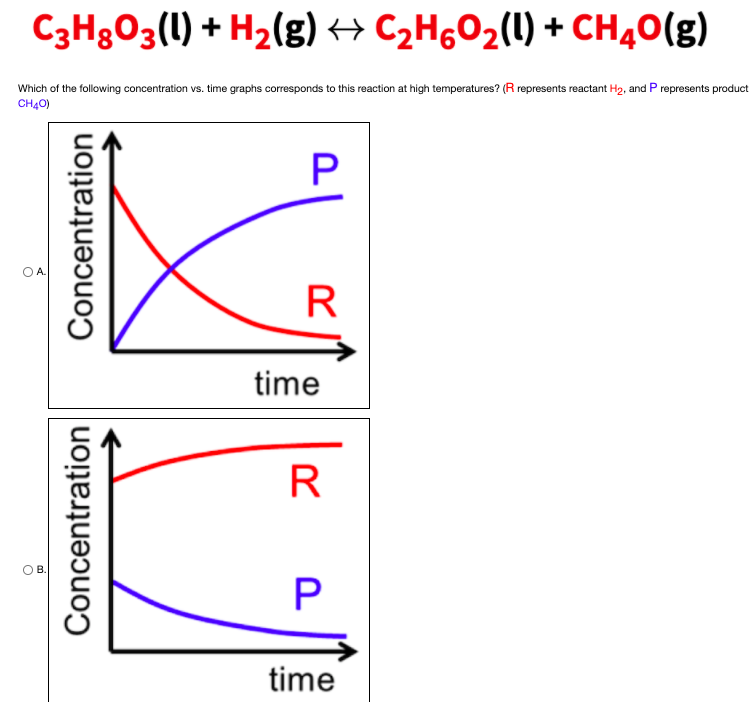
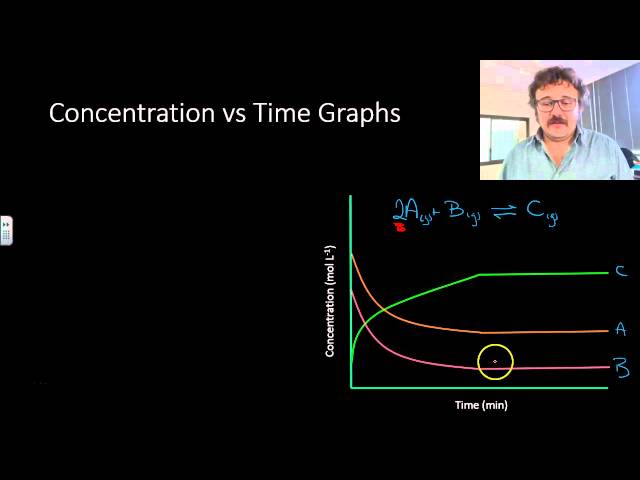


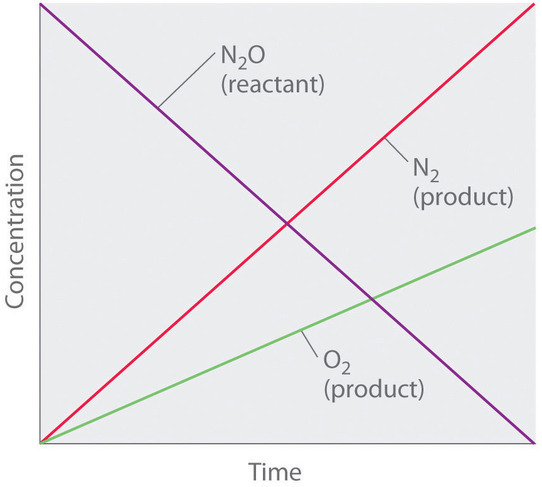
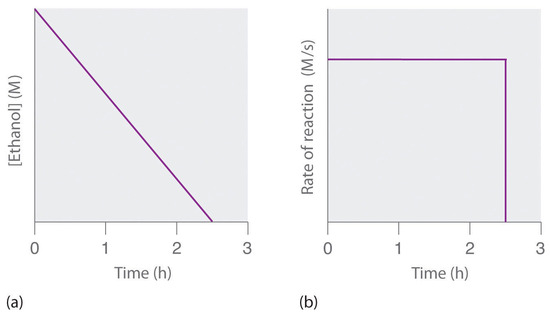

![Telugu] Drawn the graph that the concentration 'R', of the reactant a Telugu] Drawn the graph that the concentration 'R', of the reactant a](https://d10lpgp6xz60nq.cloudfront.net/physics_images/VIK_CHE_QB_C03_E01_048_S01.png)